Tirzepatide + B6 for Weight Loss
Tirzepatide mimics two gut hormones called GLP-1 (glucagon-like peptide-1) & GIP (gastric inhibitory polypeptide). When you eat, GLP-1 slows digestion and helps you feel full. It also:

Targets two hormone receptors (GLP-1 and GIP) to maximize weight loss

Is clinically proven to help lose 15-20% of body weight.

Has no hidden fees, No insurance needed.
What you need to know about Compounded Tirzepatide
What is Tirzepatide?
Tirzepatide, a GLP-1 receptor agonist, is recognized for its role in managing weight by enhancing the body’s natural ability to regulate hunger and fullness. It’s an effective method for controlling appetite and advancing toward your weight loss goals. It helps to regulate blood sugar levels, appetite, and body weight. By activating both GIP and GLP-1 receptors, Tirzepatide boosts insulin secretion, curbs glucagon production, slows down gastric emptying, and fosters feelings of fullness. This comprehensive approach aids in improving blood sugar control, shedding body weight, and potentially offering cardiovascular benefits. Clinical trials have shown promising outcomes, positioning Tirzepatide as a promising option for managing type 2 diabetes and obesity.
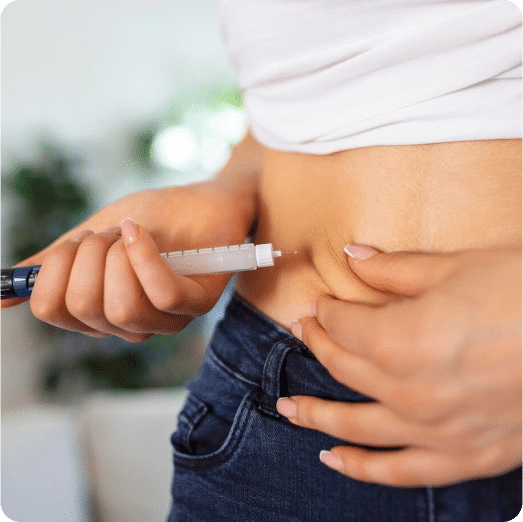
How to Take
Compounded Tirzepatide is typically injected once a week, with administration in the upper arm, thigh, or stomach. Your doctor will determine the appropriate dose for your condition and goals. Make sure to inject the medication on the same day each week and store it according to your healthcare provider’s instructions.

Benefits
- Combines two medications for better blood sugar control.
- Helps with weight loss by suppressing appetite.
- Customized to suit individual health needs.
- Reduces insulin resistance and improves glucose levels.
- Convenient once-weekly injection.

Potential Side Effects
- Nausea or upset stomach.
- Possible dizziness or fatigue.
- Headaches, particularly in the beginning.
- Digestive discomforts like bloating or diarrhea.
- Injection site redness or irritation.
- Thyroid C-Cell Tumors
- Pancreatitis
- Gallbladder Disease
- Hypoglycemia (Low Blood Sugar)
- Acute Kidney Injury
- Allergic Reactions
- Vision Changes
- Increased Heart Rate
- Mental Health Effects

Achieve Your
Optimal Health
Take Our Free 3-Min Questionnaire
Personalized Coaching Fitness & Weight Loss

Free Delivery to Your Doorstep

Lose weight effectively and keep it off
Transformative Journeys:
Real Stories of Weight Loss Wellness
solutions are empowering lives every day.

Meet Our Team

Dr. J. Chance Miller
Spenser Stokes
More Minimal Treatments
FAQs
Are there side effects of Tirzepatide?
Common side effects of Tirzepatide include:
Diarrhea & Constipation
Headaches, Nausea & Bloating
Heartburn, Stomach discomfort & Fullness
Thyroid C-Cell Tumors
Pancreatitis
Gallbladder Disease
Hypoglycemia (Low Blood Sugar)
Acute Kidney Injury
Allergic Reactions
Vision Changes
Increased Heart Rate
Mental Health Effects
How is Tirzepatide administered?
Tirzepatide is an injectable medication administered subcutaneously through the fat. The most fatty parts of the body are the abdomen, side or back of upper arms, and front of thighs.
What is the difference between Trizepatide & Semaglutide?
Tirzepatide combines two classes of medications: GLP-1 and GIP, whereas semaglutide includes just the GLP-1.
Is Tirzepatide safe?
Tirzepatide is generally considered safe for its approved use in managing diabetes; however, like all medications, it can have side effects. Its safety profile for weight loss is evaluated in ongoing clinical trials
How long can I stay on tirzepatide?
The duration of tirzepatide treatment can depend on individual health goals, response to the medication, and healthcare provider’s advice.
What is Compounded Tirzepatide with Vitamin B6?
Compounded Tirzepatide with Vitamin B6 is a specialized formulation that combines tirzepatide, the same active ingredient found in medications like Zepbound, with pyridoxine (Vitamin B6). The addition of Vitamin B6 is designed to help improve the tolerability of tirzepatide, potentially reducing the likelihood or severity of common side effects, such as nausea.
This customized approach is particularly helpful for individuals who experience intolerance to standard tirzepatide products or need a tailored solution to better fit their unique health needs. Compounded versions are available when commercially available options may not be suitable, providing more flexibility and support on your wellness journey.
All compounded medications should be prescribed by a physician and are typically dispensed through specialized compounding pharmacies, ensuring you receive the personalized care required to achieve your health goals.
How does Vitamin B6 improves Tirzepatide tolerability or reduce side effects?
Compounding Tirzepatide with vitamin B6 (Pyridoxine) can ease the initial adjustment period by reducing common side effects, particularly nausea and digestive discomfort. Vitamin B6 has long been used to support digestive health and is known to help minimize feelings of queasiness, much like its role in managing morning sickness or motion sickness. By including vitamin B6, the goal is to make your experience with Tirzepatide smoother and more comfortable as your body adapts.
What is Included in a Compounded Tirzepatide Weight Loss Journey?
With Minimal compounded Tirzepatide weight loss journey, you’ll receive a comprehensive, supportive approach designed to help you reach your health goals with confidence:
- Personalized Physician Consultation
- Convenient Home Delivery
- Ongoing Medical Oversight
- Flexible Dosing to Maximize Success
- Around-the-Clock Support
Who Is Compounded Tirzepatide with Vitamin B6 For?
Compounded Tirzepatide with Vitamin B6 is designed for individuals who have certain medical conditions and may not tolerate, or see results with, standard Tirzepatide medications like Zepbound. If you have experienced difficulty with side effects, or if your healthcare provider determines that traditional options aren’t the best fit for managing your weight or blood sugar, this compounded alternative could be considered.
Eligibility is always determined through a thorough evaluation by a licensed healthcare provider during a telehealth consultation. This ensures that every recommendation is safe, personalized, and appropriate for your unique needs. Please note that not everyone will qualify, your health history and current condition play a crucial role in the final decision.
How does compounded Tirzepatide differ from other weight loss treatment?
Unlike traditional weight loss medications that target a single hormone pathway, compounded Tirzepatide brings a unique advantage. It works by activating not just one, but two important hormone receptors in your body: GIP-1 an GIP . This dual approach amplifies your ability to curb appetite, manage fullness, and regulate blood sugar—helping you lose weight more effectively.
GIP-1 receptor agonists like Ozempic and Wegovy primarily help you feel fuller longer and reduce food cravings. Tirzepatide, however, raises the bar by also engaging the GIP pathway, which further boosts insulin sensitivity and helps your body break down fat more efficiently. The end result? Enhanced weight loss support, better blood sugar control, and a more comprehensive metabolic reset than with single-action treatments alone.
This innovative mechanism is what sets Tirzepatide apart from standard options, giving you a powerful new tool for your wellness journey.
What is the Limitation of using Tirzepatide with other medications?
Before starting tirzepatide, it’s important to understand a few key limitations.
- Tirzepatide should not be combined with other medications in the same drug class, such as additional GLP-1 receptor agonists (for example, semaglutide or liraglutide), or other tirzepatide-containing medicines.
- People with a history of pancreatitis should steer clear of tirzepatide, since its safety hasn’t been established in such cases.
- Tirzepatide is not intended for individuals with type 1 diabetes.
These precautions help ensure that tirzepatide can be used safely and effectively as part of your overall health plan.
Transform Your Health with Minimal
more fulfilling life with maximal results.